
Eq. 2.a shows that a carbon balance given by Eq. 3 is needed in the raw material
mix at a silicon yield of x if there are no losses during transportation of raw materials
and if there are no other carbon consuming reactions taking place in the process.
Fix-C = 100% • (1+x)/2
(3)
Eq. 3 shows that we need a carbon balance of 100% • (1+1)/2 = 100% if we have no
carbon losses for a silicon yield of 100%. At a silicon yield of 85%, the optimal
carbon balance is 100% • (1+0.85)/2 = 92.5%, while a silicon yield of 0% (producing
only silica according to Eq. 4 or Eq. 2.a with x=0) requires a carbon balance of 100%
• (1+0.00)/2 = 50.0%.
SiO
2
(l) + C(s) = SiO(g) + CO(g)
(4)
The important observation is that the amount of carbon required increases with 1.0%
for every 2.0% increased silicon yield. This means that more carbon must be added in
periods with high silicon yield and that the amount of carbon must be reduced if the
silicon yield drops.
Even Eq. 2 is still a simplified approach since a lot of other elements and
compounds, of which Al, Ca, Ti, S, water and volatiles are perhaps most important,
enters the process through the raw materials. However, for a discussion of the
operational aspects, Eq. 2 is just fine.
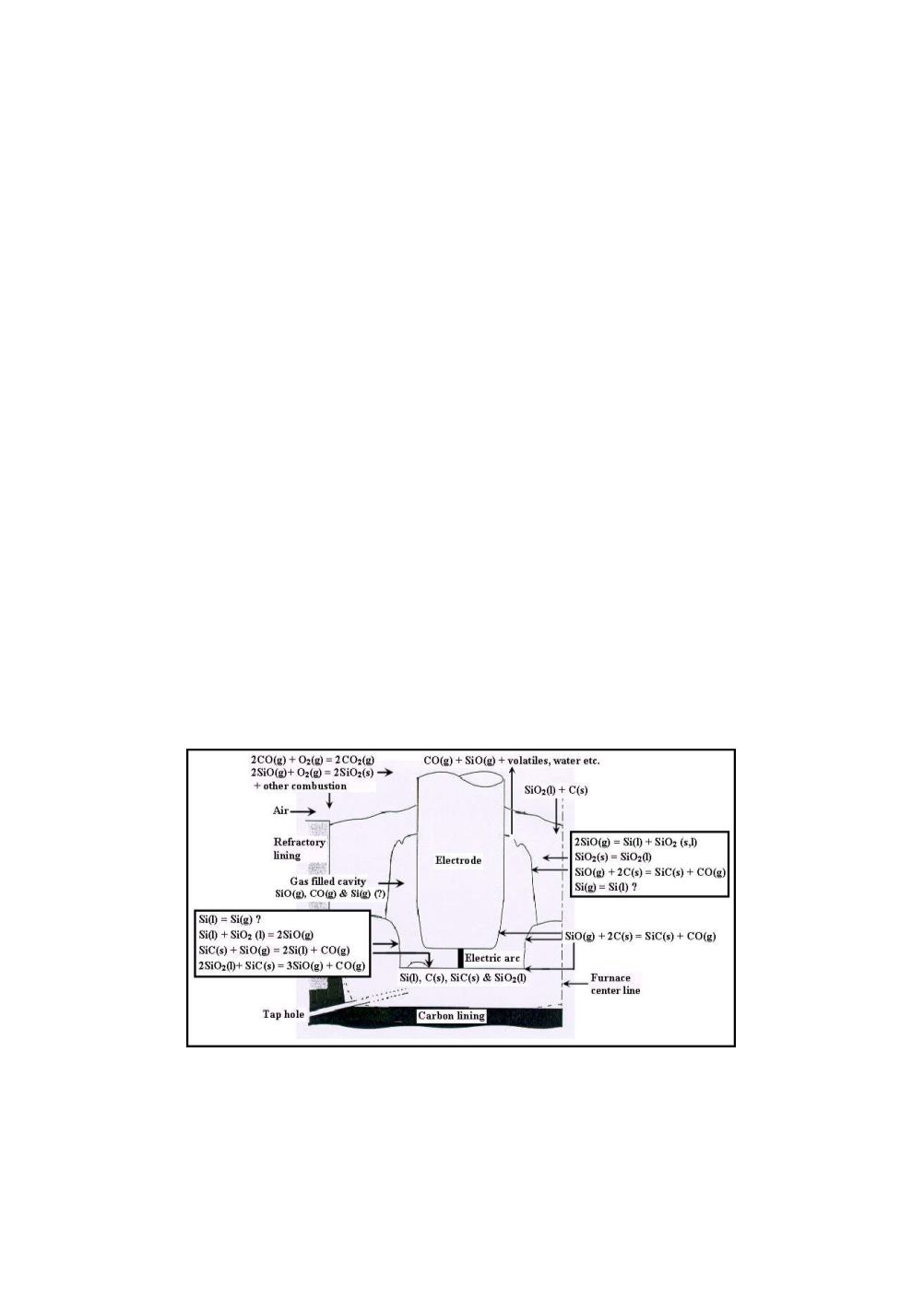
Chemical reactions in different zones
It is common to split the furnace in an upper/outer zone with mostly moderate
temperatures and a high temperature inner/lower zone surrounding the gas filled
cavity around the electrode tips. Figure 3 illustrates the conditions around one
electrode, including the most important chemical compounds and reactions in various
parts of the furnace.
Figure 3:
Schematic sketch of the area around one electrode. The figure is based on a
drawing by Schei, Tveit and Tuset [1].
High temperatures are required to produce silicon, at least 1812 C. In practice, it is
believed that the temperature around the electrode tip is in the order of 2000 C. These
67


















