
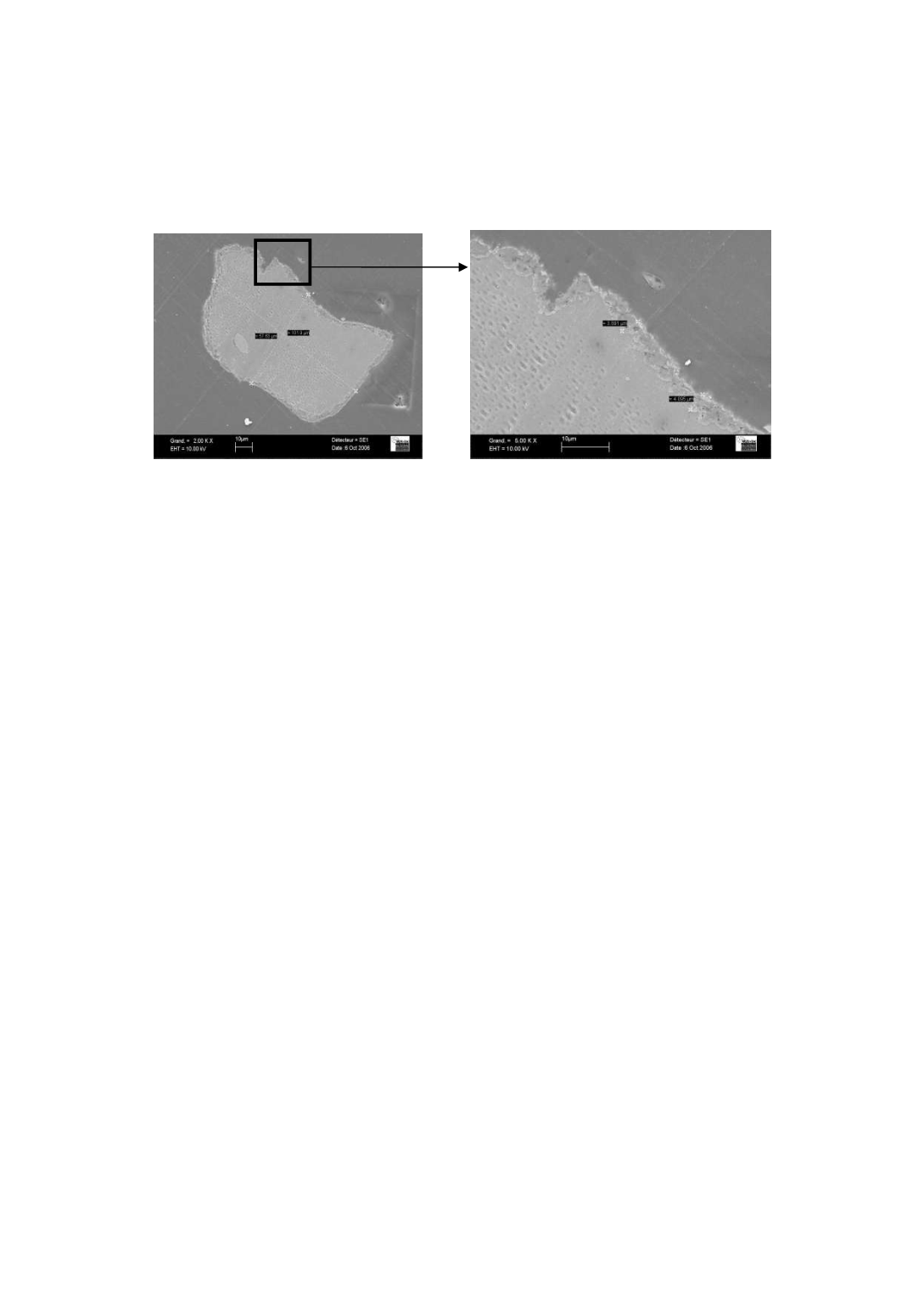
Previous work [9] has shown that the deactivation occurred together with the
formation of a thick catalyst coating layer at the surface of silicon particles, the layer
preventing contact between methyl chloride gas and silicon surface.
Figure 2
: SEM picture of silicon particle coated with catalyst, sampled during deactivation phase
Some studies describe the deactivation phenomenon. MCS reaction performed at
different Cu amounts in lab stirred bed reactor [10] indicated higher productivity and
lower initiation time with higher amount of copper, but quicker deactivation and
lower Me
2
SiCl
2
selectivity. SEM-EDX analyses indicated a rough textured surface for
deactivated masses with copper only in a shallow surface layer. Deactivation was
explained as a limited diffusion of copper into the unreacted Si surface, due to contact
between Cu discrete areas at the surface. In another study [11], main factor for the
deactivation is the enrichment of free copper. Observation of high level of carbon
deposit and high level of chlorine in the reactor containing deactivated masses could
be explained by MeCl decomposition at the surface of free copper. A mechanism was
proposed by Weber et al [12] to explain deactivation due to too high copper amounts.
In the industrial reactor, after the initiation stage, silicon is added continuously into
the FBR together with a certain amount of catalyst.
Fresh silicon particles are initiated by reaction with copper coming either from fresh
catalyst addition (same mechanism as during initiation phase) or from activated
silicon particles. This transfer of catalyst from an “old” already activated particle to a
new one will activate the fresh particle and reduce accumulation of catalyst at the
surface of the old one limiting the deactivation phenomenon described above. This
concept can be illustrated by the following scheme :
127


















